Search


EMA Guidance: Applying for Orphan Designation, IRIS Guide & Orphan Des Procedural Guidance & Forms
Recently (19 January 2023) the European Medicines Agency released an updated guidance on "IRIS guide for applicants - How to create and...

Sharan Murugan
Jan 22, 20242 min read


WHO Guidance: Ethics and Governance of Artificial Intelligence for Health
The World Health Organization (WHO) released new guidelines (18 January 2023) on ethics and governance of large multi-modal models...

Sharan Murugan
Jan 20, 20241 min read


MHRA Guidance: MAH Submission of Nitrosamine Risk Evaluation, Risk Assessment and Confirmatory Testing
Nitrosamines have become a significant concern in the pharmaceutical industry due to their potential carcinogenicity. In response to this...

Sharan Murugan
Jan 18, 20242 min read


USFDA Guidance: Requests for Reconsideration under GDUFA
The U.S. Food and Drug Administration (FDA) Center for Drug Evaluation and Research and Center for Biologics Evaluation and Research...

Sharan Murugan
Jan 13, 20242 min read


EMA: Guidance on Paediatric Submissions & Scientific Guidelines with SmPC Recommendations
Developing drugs specifically for paediatric use comes with its own set of challenges, including ethical considerations, appropriate...

Sharan Murugan
Jan 13, 20241 min read


UK MHRA: Guidance on 150-Day Assessment for National Applications for Medicines
Earlier today (05 January 2024) UK's Medicines and Healthcare Products Regulatory Agency (MHRA) released updated guidance on "150-day...

Sharan Murugan
Jan 5, 20242 min read


UK MHRA: Guidance on Operational Information Sharing
Earlier today (02 January 2024) UK's Medicines and Healthcare Products Regulatory Agency (MHRA) released updated guidance on "Operational...

Sharan Murugan
Jan 2, 20241 min read


USFDA : CMC Guidance Reformulating Drug Products That Contain Carbomers & Potency Assurance for Cellular & Gene Therapy Products
The U.S. Food and Drug Administration (FDA) Center for Drug Evaluation and Research and Center for Biologics Evaluation and Research...

Sharan Murugan
Dec 30, 20232 min read


USFDA Guidance: Considerations, Master Protocols for the Development of Drugs and Biological Products
The U.S. Food and Drug Administration (FDA) Center for Drug Evaluation and Research and Center for Biologics Evaluation and Research...

Sharan Murugan
Dec 27, 20232 min read


USFDA Guidance: Digital Health Technologies for Remote Data Acquisition & Data Standards for Drug and Biological Product Submissions and Real-World Data
Earlier this week the the U.S. Food and Drug Administration (FDA) released multiple guidances that is related to Digital Technologies and...

Sharan Murugan
Dec 27, 20232 min read


UK MHRA: Guidance on Submitting Changes to Labelling and Patient Information Leaflets
On 22nd December 2023, the Medicines and Healthcare Products Regulatory Agency (MHRA) updated its guidance on Medicines "Submitting...

Sharan Murugan
Dec 27, 20232 min read


Singapore's HSA: Post-Industry Consultation Updates on eCTD Implementation in Singapore
On 11th December 2023, Singapore's Health Sciences Authority (HSA) released an update "Post-Industry Consultation Updates on Electronic...

Sharan Murugan
Dec 11, 20231 min read


UK MHRA Guidance: Apply for a Parallel Import Licence
Earlier today (05 December 2023) Medicines and Healthcare Products Regulatory Agency (MHRA) updated its guidance on Medicines: "Apply for...

Sharan Murugan
Dec 5, 20232 min read


Health Canada: Guidance on Validation rules for Regulatory Transactions in the eCTD Format
Health Canada recently published an updated guidance on "Validation rules for regulatory transactions provided to Health Canada in the...

Sharan Murugan
Nov 30, 20231 min read


USFDA Guidance Q&A: Translation of Good Laboratory Practice Study Reports
On 21 November,2023 the USFDA issued updated guidance on "Translation of Good Laboratory Practice Study Reports: Questions and Answers"...

Sharan Murugan
Nov 24, 20232 min read


UK MHRA: Guidance on Change of Ownership: Marketing Authorisation Process
Last Friday (17 November 2023) UK's Medicines and Healthcare Products Regulatory Agency (MHRA) released updated guidance on "Medicines...

Sharan Murugan
Nov 18, 20232 min read

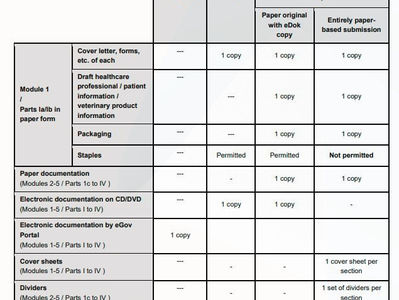
Switzerland's SwissMedic: Guidance on Formal Requirements
On 15 November 2023, Switzerland's Swissmedic released an updated guidance on "Formal Requirements" which serves as a resource for...

Sharan Murugan
Nov 15, 20232 min read


USFDA Guidance: Real-Time Oncology Review (RTOR)
The U.S. Food and Drug Administration (FDA) Oncology Center of Excellence (OCE) released guidance earlier this week (07 November 2023)...

Sharan Murugan
Nov 11, 20232 min read


MHRA Guidance: Common Issues Identified during Clinical Trial Applications
Yesterday (06 November 2023) the UK's Medicines and Healthcare Products Regulatory Agency (MHRA) released updated guidance on "Common...

Sharan Murugan
Nov 7, 20231 min read


USFDA Guidance: Submitting Patient-Reported Outcome Data, Clinical Trial Datasets & Documentation
Today (06 November, 2023) the U.S. Food and Drug Administration (FDA) Center for Drug Evaluation and Research (CDER) released two final...

Sharan Murugan
Nov 6, 20232 min read
