Search

EMA Guidance: Electronic Submission of Medicinal Product Data under IDMP (Chapter 3 Explained)
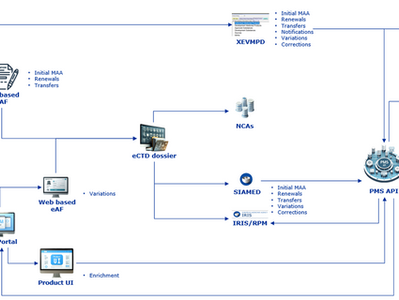
The European Medicines Agency (EMA) continues to advance data standardisation and digital transformation in regulatory processes through the implementation of ISO IDMP standards and Product Management Services (PMS). The guidance Process for the electronic submission of medicinal product information – Chapter 3 (Version 3.5, updated 20 April 2026) provides detailed instructions on how medicinal product data should be submitted, managed, and maintained electronically throughou

Sharan Murugan
3 days ago3 min read
