Search


EMA GVP Module VII – Explanatory Note (Rev. 4): Periodic Safety Update Report (PSUR) Single Assessment
The European Medicines Agency (EMA) has issued Revision 4 of the "Explanatory Note to Good Pharmacovigilance Practices (GVP) Module VII – Periodic Safety Update Report" prepared by the Human Medicines Evaluation Division. This revision addresses practical challenges encountered during the PSUR Single Assessment (PSUSA) process, particularly for nationally authorised medicinal products (NAPs), and serves as the basis for the forthcoming update of GVP Module VII. Purpose of the

Sharan Murugan
2 days ago3 min read

USFDA Guidance: Continuous Glucose Monitoring Data in Clinical Trials and Postapproval Pregnancy Safety Studies
The U.S. Food and Drug Administration (FDA) continues to modernize clinical research and postmarketing safety monitoring through new guidance documents focused on digital health technologies and real-world safety evidence. In May 2026, FDA published two important guidance documents: Submitting Continuous Glucose Monitoring Data in Clinical Trials Postapproval Pregnancy Safety Studies Guidance on Continuous Glucose Monitoring (CGM) Data in Clinical Trials What is Continuous Gl

Sharan Murugan
May 103 min read


USFDA Guidance: CMC Flexibilities to Accelerate Human Cellular and Gene Therapy Development
Human cellular and gene therapy (CGT) products are transforming modern medicine by offering innovative treatments for serious and life-threatening diseases such as cancer, genetic disorders, and chronic illnesses. However, the development and manufacturing of these therapies are often highly complex due to personalized production processes, advanced technologies, limited patient populations, and short product shelf-lives. To support innovation while maintaining product qualit

Sharan Murugan
May 104 min read


USFDA’s Press Release: Real-Time Clinical Trials and the Next Era of Drug Development
The traditional clinical trial model has long been associated with delays in data reporting, lengthy timelines, and inefficiencies in decision-making. Recognizing these challenges, the U.S. Food and Drug Administration (FDA) has taken a transformative step toward modernizing clinical research. This press announcement was released on April 28, 2026, marking a significant milestone in the evolution of clinical trials. The FDA introduced major steps to advance the implementation

Sharan Murugan
May 43 min read


EMA’s Guidance on Computerised Systems: Ensuring Data Integrity in Clinical Trials
The integrity, reliability, and robustness of clinical trial data are fundamental to regulatory decision-making, particularly for marketing authorisation applications (MAAs). With the increasing reliance on computerised systems such as electronic case report forms (eCRFs), electronic patient-reported outcomes (ePROs), safety databases, interactive response technologies (eIRT), and clinical trial management systems (CTMS), the role of validated digital systems has become centr

Sharan Murugan
May 43 min read


Singapore HSA Guidance: Post-Marketing Vigilance Requirements for Therapeutic Products and CTGTPs in 2026
The Singapore Health Sciences Authority (HSA) has released Version 6 of its updated guidance document titled “Post-Marketing Vigilance Requirements for Therapeutic Products and Cell, Tissue and Gene Therapy Products (CTGTP)”, effective from 1 April 2026. The updated guidance provides important clarification on pharmacovigilance obligations, adverse event reporting, risk management plans (RMPs), and post-registration safety responsibilities for companies operating in Singapore

Sharan Murugan
Apr 303 min read


Philippines FDA Draft Guidance: Adoption and Implementation of Good Registration Management (GRM), Good Review Practice (GRevP), and Good Submission Practice (GSubP) for Pharmaceutical Products
The Philippines Food and Drug Administration (FDA) is advancing its regulatory framework by adopting internationally aligned practices to improve the quality, transparency, and efficiency of pharmaceutical product regulation. The draft circular Adoption and Implementation of Good Registration Management (GRM), Good Review Practice (GRevP), and Good Submission Practice (GSubP) for Pharmaceutical Products introduces a structured and integrated approach to both regulatory submi

Sharan Murugan
Apr 253 min read


TGA Guidance: Mandatory Reporting of Medicine Shortages and Discontinuations
Medicine shortages and discontinuations are a growing global concern, and Australia has established a robust regulatory framework to ensure early detection, transparency, and risk mitigation. The guidance Reporting a shortage or discontinuation of a medicine you supply (updated 13 April 2026) outlines the mandatory obligations for sponsors to report medicine shortages and discontinuations to the Therapeutic Goods Administration (TGA). A medicine shortage occurs when supply is

Sharan Murugan
Apr 252 min read

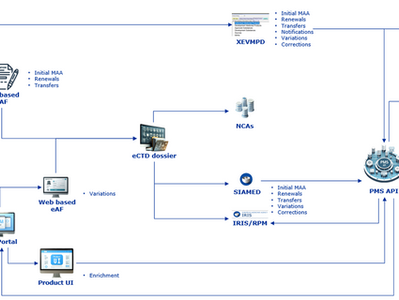
EMA Guidance: Electronic Submission of Medicinal Product Data under IDMP (Chapter 3 Explained)
The European Medicines Agency (EMA) continues to advance data standardisation and digital transformation in regulatory processes through the implementation of ISO IDMP standards and Product Management Services (PMS). The guidance Process for the electronic submission of medicinal product information – Chapter 3 (Version 3.5, updated 20 April 2026) provides detailed instructions on how medicinal product data should be submitted, managed, and maintained electronically throughou

Sharan Murugan
Apr 253 min read


EMA & HC Guidance: Clinical Data Publication (CDP) – EMA Policy 0070 Work-Share with Health Canada
The European Medicines Agency (EMA) continues to strengthen transparency, data sharing, and international regulatory collaboration through its Clinical Data Publication (CDP) framework. A key development in this area is the introduction of a work-share initiative with Health Canada (HC) under Policy 0070, aimed at improving efficiency while maintaining robust safeguards for sensitive information. The guidance Clinical Data Publication (CDP): Questions and Answers on EMA Polic

Sharan Murugan
Apr 253 min read


UK MHRA Guidance: Compliance with ICH E6 GCP in UK, Quality and Risk proportionality, & Clinical Investigations for Medical Devices
The UK clinical trial regulatory framework is undergoing a major transformation with the implementation of updated ICH E6 (R3) Good Clinical Practice (GCP) principles, alongside new guidance on quality, risk proportionality, and clinical investigations. The guidance Clinical trials for medicines: Compliance with ICH E6 Good Clinical Practice (GCP) in the United Kingdom, Clinical trials for medicines: guidance on quality and risk proportionality, and Clinical investigations fo

Sharan Murugan
Apr 253 min read


USFDA Guidance: Safety Assessment of Genome Editing in Human Gene Therapy Products Using NGS and Expanded Access to Investigational Drugs for Treatment Use-Q&A
The regulatory landscape for advanced therapies and patient access in the United States continues to evolve, with the FDA issuing updated guidances in April 2026 that address both cutting-edge genome editing safety and patient access to investigational treatments . Together, these guidances highlight the balance between innovation, safety evaluation, and ethical access to therapies across the product lifecycle. The guidance Safety Assessment of Genome Editing in Human Gene

Sharan Murugan
Apr 203 min read


EMA Guidance: Electronic Submission of IMP Information in XEVMPD
The Guidance on the electronic submission of information on investigational medicinal products for human use in the Extended EudraVigilance medicinal product dictionary (XEVMPD) was updated on 13 April 2026 (Version 1.5) , providing detailed procedural and technical instructions for sponsors submitting clinical trial product data. Introduction This guidance provides consolidated instructions for sponsors on how to electronically submit information on investigational medici

Sharan Murugan
Apr 203 min read


EMA Guidance: Orphan Medicinal Product Designation & Paediatric Applications
The European regulatory landscape continues to evolve with a strong focus on supporting innovation in rare diseases and ensuring safe, evidence-based development of medicines for children . The European Medicines Agency (EMA) has issued two key procedural guidelines: Procedural advice for orphan medicinal product designation – Guidance for sponsors Procedural advice on paediatric applications – Guidance for applicants together, establish a comprehensive regulatory framewo

Sharan Murugan
Apr 202 min read


UK Guidance: Clinical trials for medicines: Guidance on Compliance with ICH E6 Good Clinical Practice (GCP) in the United Kingdom
The UK regulatory framework for clinical trials is undergoing a significant transition with the adoption of updated Good Clinical Practice (GCP) standards aligned with international expectations. The Medicines and Healthcare products Regulatory Agency (MHRA) is reinforcing compliance with ICH E6 (R3) to ensure high standards of participant safety, data integrity, and efficient trial conduct. The guidance Clinical trials for medicines: Guidance on compliance with ICH E6 Good

Sharan Murugan
Apr 163 min read


UK MHRA Guidance: National Assessment Procedure for Medicines
The UK regulatory system for medicines continues to advance towards faster access, aligned decision-making, and patient-focused innovation . The Medicines and Healthcare products Regulatory Agency (MHRA) is strengthening its national procedures to ensure efficient evaluation of medicines while improving coordination with health technology assessment bodies. The guidance National assessment procedure for medicines was updated on 9 April 2026 , including the addition of a webi

Sharan Murugan
Apr 122 min read


USFDA Guidance: Incorporating Voluntary Patient Preference Information over the Total Product Life Cycle
The guidance Incorporating Voluntary Patient Preference Information over the Total Product Life Cycle was issued on 30 March 2026 , updating and superseding the earlier 2016 guidance, and providing comprehensive recommendations on how patient preferences can inform benefit-risk assessments. The FDA recognizes that patients living with diseases or conditions develop first-hand insights into treatment benefits and risks. Their perspectives can play a critical role in evaluatin

Sharan Murugan
Apr 52 min read


UK MHRA: Guidance on Environmental Risk Assessments in medicines
The UK regulatory landscape for medicines is increasingly evolving to integrate environmental protection alongside public health safety, and recently, on 1 April 2026, the UK MHRA updated its guidance on " Environmental Risk Assessments in medicines " . A key component of this approach is the requirement for Environmental Risk Assessments (ERA), ensuring that medicinal products do not pose unintended risks to ecosystems during their lifecycle. Background Environmental Risk

Sharan Murugan
Apr 52 min read


India CDSCO Notice: Draft Notification on Post-Approval Changes (PAC) in Drugs
India’s Central Drugs Standard Control Organization (CDSCO) has released a draft notification dated 09 March 2026 proposing amendments to the Drugs Rules, 1945 to establish a structured framework for managing post-approval manufacturing changes. This draft notification introduces clear regulatory requirements for notifying authorities when manufacturers make changes to manufacturing processes, excipients, packaging, shelf life, specifications, testing procedures, or docume

Sharan Murugan
Mar 212 min read

EDQM Guidance: Reliance-Based and Fast-Track Assessment of CEP Applications
This guidance , published in March 2026 by the European Directorate for the Quality of Medicines & HealthCare (EDQM) , introduces new approaches for accelerating the assessment of Certificates of Suitability (CEP) applications. It focuses on two key pathways: Fast-track assessment Reliance-based assessment Both approaches aim to improve regulatory efficiency, reduce delays, and ensure timely access to medicines. CEP procedures are essential for demonstrating that a substance

Sharan Murugan
Mar 212 min read
