Search

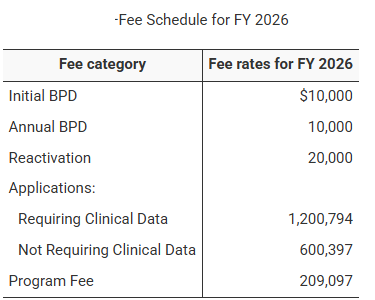
USFDA Announcement: Biosimilar User Fee Rates for Fiscal Year 2026 (October 1, 2025 – September 30, 2026)
On 30 July 2025, the U.S. Food and Drug Administration (FDA) officially published the Biosimilar User Fee Rates for Fiscal Year (FY) 2026...

Sharan Murugan
Aug 2, 20252 min read


USFDA Guidance: Formal Meetings Between the FDA and Sponsors or Applicants of BsUFA Products
In July 2025, the U.S. Food and Drug Administration (FDA) published its updated guidance for industry titled “ Formal Meetings Between...

Sharan Murugan
Jul 19, 20253 min read


USFDA Guidance: User Fee Programs: GDUFA, BSUFA, OMOR, and MDUFA
On July 31, 2024 the U.S. Food and Drug Administration (FDA) recently published in the Federal Register notice (FRN) the User Fee Rates...

Sharan Murugan
Jul 31, 20243 min read


USFDA Guidance: Formal Meetings Between the FDA and Sponsors or Applicants of BsUFA Products
Today (11 August 2023) the United States Food & Drug Administration released a draft guidance “Formal Meetings Between the FDA and...

Sharan Murugan
Aug 10, 20232 min read


USFDA Guidance: Assessing User Fees -BsUFA III: Biosimilar User Fee Amendments of 2022
Today (31 July 2023) the United States Food & Drug Administration's Center for Biologics Evaluation and Research and Center for Drug...

Sharan Murugan
Aug 1, 20232 min read


FY2022 User Fee Table -USFDA
The User Fee programs help the Food and Drug Administration (FDA) to fulfill its mission of protecting public health and accelerating...

Sharan Murugan
Aug 13, 20212 min read
