Search


EMA GVP Module VII – Explanatory Note (Rev. 4): Periodic Safety Update Report (PSUR) Single Assessment
The European Medicines Agency (EMA) has issued Revision 4 of the "Explanatory Note to Good Pharmacovigilance Practices (GVP) Module VII – Periodic Safety Update Report" prepared by the Human Medicines Evaluation Division. This revision addresses practical challenges encountered during the PSUR Single Assessment (PSUSA) process, particularly for nationally authorised medicinal products (NAPs), and serves as the basis for the forthcoming update of GVP Module VII. Purpose of the

Sharan Murugan
6 days ago3 min read

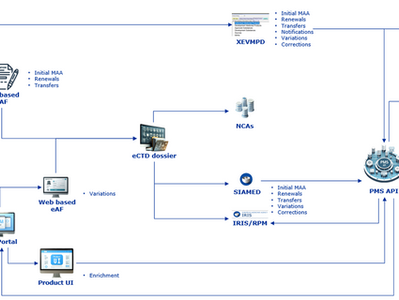
EMA Guidance: Electronic Submission of Medicinal Product Data under IDMP (Chapter 3 Explained)
The European Medicines Agency (EMA) continues to advance data standardisation and digital transformation in regulatory processes through the implementation of ISO IDMP standards and Product Management Services (PMS). The guidance Process for the electronic submission of medicinal product information – Chapter 3 (Version 3.5, updated 20 April 2026) provides detailed instructions on how medicinal product data should be submitted, managed, and maintained electronically throughou

Sharan Murugan
Apr 253 min read


EMA & HC Guidance: Clinical Data Publication (CDP) – EMA Policy 0070 Work-Share with Health Canada
The European Medicines Agency (EMA) continues to strengthen transparency, data sharing, and international regulatory collaboration through its Clinical Data Publication (CDP) framework. A key development in this area is the introduction of a work-share initiative with Health Canada (HC) under Policy 0070, aimed at improving efficiency while maintaining robust safeguards for sensitive information. The guidance Clinical Data Publication (CDP): Questions and Answers on EMA Polic

Sharan Murugan
Apr 253 min read


EMA Guidance: Electronic Submission of IMP Information in XEVMPD
The Guidance on the electronic submission of information on investigational medicinal products for human use in the Extended EudraVigilance medicinal product dictionary (XEVMPD) was updated on 13 April 2026 (Version 1.5) , providing detailed procedural and technical instructions for sponsors submitting clinical trial product data. Introduction This guidance provides consolidated instructions for sponsors on how to electronically submit information on investigational medici

Sharan Murugan
Apr 203 min read


EMA Guidance: Orphan Medicinal Product Designation & Paediatric Applications
The European regulatory landscape continues to evolve with a strong focus on supporting innovation in rare diseases and ensuring safe, evidence-based development of medicines for children . The European Medicines Agency (EMA) has issued two key procedural guidelines: Procedural advice for orphan medicinal product designation – Guidance for sponsors Procedural advice on paediatric applications – Guidance for applicants together, establish a comprehensive regulatory framewo

Sharan Murugan
Apr 202 min read


EMA Notice: European Medicines Agency’s Data Protection Notice for the implementation of the Instrument for Pre-accession Assistance (IPA) programme
The European regulatory framework continues to strengthen data protection and transparency in its international collaborations, particularly when engaging with EU candidate countries. As part of this effort, the European Medicines Agency (EMA) has established clear guidelines on how personal data is processed within cooperation programmes. The guidance European Medicines Agency’s Data Protection Notice for the implementation of the Instrument for Pre-accession Assistance (I

Sharan Murugan
Apr 82 min read


EMA Med.Dev Guidance: Guide to Requesting Advice from Expert Panels for Orphan Medical Devices
The guidance Guide to manufacturers and notified bodies on the procedure for requesting advice from expert panels for orphan medical devices was published on 31 March 2026 , outlining the procedural framework and expectations for applicants seeking scientific and regulatory advice. Orphan medical devices are those intended for the diagnosis, prevention, or treatment of rare conditions, where limited patient populations create challenges in generating extensive clinical evi

Sharan Murugan
Apr 53 min read


EMA Guidance: Chemistry of Active Substances and GMP for Additive Manufacturing (3DP)
The European Medicines Agency (EMA) has introduced two significant guidance updates in 2026, focusing on active substance chemistry requirements and the emerging use of additive manufacturing (3D printing) in pharmaceuticals . These updates aim to strengthen quality, control strategies, and innovation , while ensuring that pharmaceutical development and manufacturing remain scientifically robust and patient-focused. Guidance: Chemistry of Active Substances Scope and Regulato

Sharan Murugan
Mar 294 min read


EMA Guidance: Implementation of ICH E2D(R1) for Post-Approval Safety Reporting and the EU Network Data & AI Strategy
Regulatory authorities across the European Union continue to strengthen pharmacovigilance systems and data-driven regulatory decision-making. Two important EMA documents highlight these developments: the EU implementation strategy for the ICH E2D(R1) guideline on post-approval safety data and the Network Data Steering Group (NDSG) Workplan 2026–2028 on data and artificial intelligence in medicines regulation. Guidance: Network Data Steering Group Workplan 2026–2028: Data and

Sharan Murugan
Mar 154 min read


USFDA Guidance: Use of Artificial Intelligence To Support Regulatory Decision & Guiding Principles of Good AI Practice in Drug Development
Artificial Intelligence (AI) is no longer a future concept in pharmaceutical research—it is already influencing how drugs and biological products are discovered, developed, manufactured, and monitored throughout their lifecycle. However, when AI outputs are used to support regulatory decisions , questions of trust, transparency, and accountability become critical. Recognising this, global regulators have begun defining expectations for the responsible use of AI in drug devel

Sharan Murugan
Jan 313 min read


EMA Overview: European Shortages Monitoring Platform (ESMP): How the EU Monitors, Prevents, and Manages Medicine Shortages
Medicine shortages can have a direct and serious impact on patient care, healthcare systems, and public health across Europe. To strengthen coordination, improve visibility, and enable early action, the European Medicines Agency (EMA) has established the European Shortages Monitoring Platform (ESMP) . The ESMP is a central digital platform designed to support the prevention, identification, monitoring, and management of shortages of medicinal products across the EU and EEA.

Sharan Murugan
Dec 20, 20253 min read


EMA Draft Guideline on Quality of Radiopharmaceuticals
Radiopharmaceuticals present unique quality challenges due to their radioactive nature, short shelf lives, and frequent need for on-site or near-patient preparation. To reflect scientific and technological advances since the original 2007 guideline, the European Medicines Agency has released the draft Guideline on quality of radiopharmaceuticals – Revision 2 for public consultation. Adopted by CHMP in December 2025, this revision provides updated expectations for quality doc

Sharan Murugan
Dec 13, 20252 min read


EMA Guideline on the Development, Manufacture of Synthetic Peptides and Good Pharmacogenomic Practice
Modern medicines development increasingly sits at the intersection of advanced chemistry and genomic science. On one hand, synthetic peptides represent a fast-growing therapeutic class that bridges small molecules and biologics. On the other, pharmacogenomics is reshaping how medicines are developed, evaluated, and ultimately used in patients. Recognising these parallel advances, the European Medicines Agency (EMA) has recently published two key scientific documents: the Guid

Sharan Murugan
Dec 13, 20253 min read


EMA Guideline on Stability Testing for Applications for Variations to a Marketing Authorisation
Post-authorisation changes are an inevitable part of medicinal product lifecycle management, and stability data play a critical role in demonstrating that such changes do not compromise product quality, safety, or efficacy. To harmonise expectations across the EU, the European Medicines Agency has issued Revision 3 of the Guideline on stability testing for applications for variations to a marketing authorisation . Adopted in December 2025 and effective from January 2026, this

Sharan Murugan
Dec 13, 20252 min read


EMA Q&A Guidance: Updated Classification of (Post-Authorisation) Changes
Regulatory teams responsible for lifecycle management know that post-authorisation changes can quickly become complex, especially as products mature, manufacturing processes evolve, and emerging scientific data require frequent updates to quality and clinical documentation. To support consistent interpretation of EU Variation Regulation (EC) No 1234/2008, the European Medicines Agency (EMA) maintains an extensive Q&A document titled " Classification of Changes " —a practical

Sharan Murugan
Dec 7, 20254 min read


EMA Guidance: ETF Scientific Advice that facilitates Clinical Trial Authorisations (SA-CTA) and 2025–2027 IRIS Roadmap
The European Medicines Agency (EMA) recently published new guidance titled “ Guidance for applicants: the ETF Scientific Advice that facilitates Clinical Trial Authorisations (SA-CTA ) ”, outlining how sponsors and applicants can benefit from harmonised scientific advice to accelerate clinical trial authorisation across the EU. 1. What the SA-CTA Scientific Advice Is The guidance explains that the SA-CTA scientific advice is a specialised type of EMA advice developed under th

Sharan Murugan
Nov 23, 20254 min read


EMA’s New Draft Guideline: Non-Inferiority and Equivalence Comparisons in Clinical Trials
The European Medicines Agency (EMA) has released a major draft guideline " Non-Inferiority and Equivalence Comparisons in Clinical Trials " that updates how non-inferiority and equivalence comparisons should be designed, justified, and analysed in confirmatory clinical trials. This draft replaces two earlier documents: Guideline on the choice of the non-inferiority margin (2005), and Points to consider on switching between superiority and non-inferiority (2000). The new gu

Sharan Murugan
Nov 17, 20252 min read


EMA Guidance: Regulating Innovation in Phage Therapy and Device–Drug Combinations
In October 2025, the European Medicines Agency (EMA) released two landmark drafts for public consultation. These documents collectively signal the Agency’s evolving regulatory stance toward precision biologics such as bacteriophage therapy and innovative device–drug integration approaches to simplify clinical bridging for biologics delivered subcutaneously. 1. Quality Guidance for Phage Therapy Medicinal Products (PTMPs) The EMA draft guideline on phage therapy quality (Oct

Sharan Murugan
Oct 26, 20252 min read


EMA Guidance: European Commission’s (EC) New Variations Guidelines
On 22 September 2025 , the European Commission published new variations guidelines in the Official Journal of the European Union. These...

Sharan Murugan
Sep 22, 20252 min read


EMA Publishes First EU eCTD v4.0 Validation Criteria and Updated Controlled Vocabularies
In 1st & 8th August 2025, the European Medicines Agency (EMA) announced two major milestones in the transition to electronic Common...

Sharan Murugan
Aug 10, 20252 min read
