Search

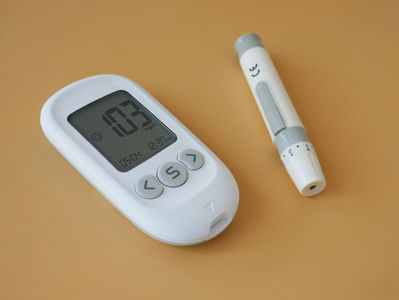
USFDA Guidance: Continuous Glucose Monitoring Data in Clinical Trials and Postapproval Pregnancy Safety Studies
The U.S. Food and Drug Administration (FDA) continues to modernize clinical research and postmarketing safety monitoring through new guidance documents focused on digital health technologies and real-world safety evidence. In May 2026, FDA published two important guidance documents: Submitting Continuous Glucose Monitoring Data in Clinical Trials Postapproval Pregnancy Safety Studies Guidance on Continuous Glucose Monitoring (CGM) Data in Clinical Trials What is Continuous Gl

Sharan Murugan
2 days ago3 min read


USFDA Guidance: For Patient-Matched Guides Used in Orthopedic Implants
Patient-matched medical devices are becoming increasingly important in modern orthopedic surgery. These devices are designed specifically for individual patients using imaging data such as CT scans or MRI scans. The goal is to help surgeons position orthopedic implants more accurately during surgery and improve overall surgical planning. To provide clearer regulatory expectations for manufacturers, the U.S. Food and Drug Administration (FDA) published the guidance titled “Pat

Sharan Murugan
2 days ago4 min read


USFDA Press Release: AI Capabilities with Elsa 4.0 and New HALO Data Platform
Artificial intelligence (AI) is rapidly becoming an important part of healthcare, drug development, and regulatory operations. To modernize its internal systems and improve operational efficiency, the U.S. Food and Drug Administration (FDA) announced major upgrades to its AI infrastructure and data management capabilities. On May 6, 2026, the FDA announced the launch of Elsa 4.0, an upgraded version of its internal AI tool, along with the completion of a large-scale data plat

Sharan Murugan
2 days ago3 min read


USFDA Guidance: CMC Flexibilities to Accelerate Human Cellular and Gene Therapy Development
Human cellular and gene therapy (CGT) products are transforming modern medicine by offering innovative treatments for serious and life-threatening diseases such as cancer, genetic disorders, and chronic illnesses. However, the development and manufacturing of these therapies are often highly complex due to personalized production processes, advanced technologies, limited patient populations, and short product shelf-lives. To support innovation while maintaining product qualit

Sharan Murugan
2 days ago4 min read


European Commission: Introduces New Rules for Notified Bodies Under MDR and IVDR
The European Union has taken another major step toward strengthening the efficiency, predictability, and consistency of medical device and in vitro diagnostic (IVD) certification processes. On 4 May 2026, the European Commission adopted Commission Implementing Regulation (EU) 2026/977, which was subsequently published in the Official Journal of the European Union on 5 May 2026. The regulation establishes uniform quality management and procedural requirements for conformity as

Sharan Murugan
5 days ago3 min read


EMA’s Guidance on Computerised Systems: Ensuring Data Integrity in Clinical Trials
The integrity, reliability, and robustness of clinical trial data are fundamental to regulatory decision-making, particularly for marketing authorisation applications (MAAs). With the increasing reliance on computerised systems such as electronic case report forms (eCRFs), electronic patient-reported outcomes (ePROs), safety databases, interactive response technologies (eIRT), and clinical trial management systems (CTMS), the role of validated digital systems has become centr

Sharan Murugan
May 43 min read
