Search

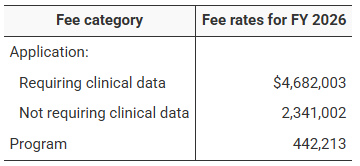
USFDA FDA User Fee Rates for FY 2026: A Comparative Overview for Prescription Drugs, Medical Devices, Generics, and Outsourcing Facilities
The U.S. Food and Drug Administration (FDA) has announced On 30 July 2025 , the official user fee rates across its major healthcare...

Sharan Murugan
Aug 2, 20252 min read


USFDA Guidance: User Fee Programs: GDUFA, BSUFA, OMOR, and MDUFA
On July 31, 2024 the U.S. Food and Drug Administration (FDA) recently published in the Federal Register notice (FRN) the User Fee Rates...

Sharan Murugan
Jul 31, 20243 min read


USFDA Guidance: Four Finalized User Fees and Refunds related Medical Devices Guidelines
USFDA's Center for Devices and Radiological Health and Center for Biologics Evaluation and Research finalized and released four Medical...

Sharan Murugan
Oct 6, 20221 min read


FY2022 User Fee Table -USFDA
The User Fee programs help the Food and Drug Administration (FDA) to fulfill its mission of protecting public health and accelerating...

Sharan Murugan
Aug 13, 20212 min read
