Guideline for Registration of Drug-Medical Device & Med.Dev-Drug Combo Products - MALAYSIA -NPRA
- Sharan Murugan

- Nov 7, 2021
- 1 min read
Drug-Medical Device Combination Product (DMDCP): Primary mode of action is based on pharmacological, immunological or metabolic action in/on the body where NPRA is the primary agency of the combination product.
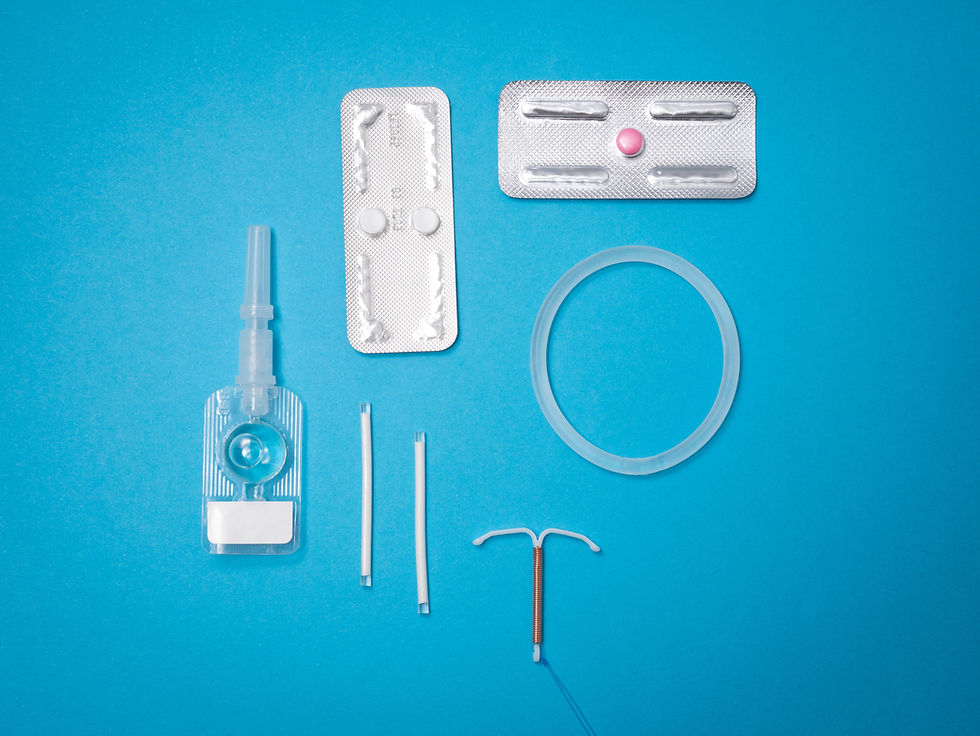
This present guideline serves as guidance for the submission of a registration application of drug-medical device/ medical device-drug combination products, and change/variation application.
Drug-medical device/ medical device-drug combination products are regulated according to the classification whether as drug or medical device based on the primary mode of action (PMOA).
The scope of this guideline includes information relating to dossier requirements, procedures for submission of combination products registration and change/variation application. Applicants shall familiarize with the contents of this guidance document and the governing legislations before they submit registration applications.
Check out this guideline for more detailed information



Comments