Search


India CDSCO: Launching of National Single Window System (NSWS) Portal
To address the challenges in the management of medical devices, India's Central Drugs Standard Control Organization (CDSCO) has...

Sharan Murugan
Jan 2, 20241 min read


Saudi Arabia’s (SFDA): Requirements for Clinical Trials of Medical Devices
The Saudi Food and Drug Authority (SFDA) issued a comprehensive updated guidance titled "Requirements for Clinical Trials of Medical...

Sharan Murugan
Dec 30, 20231 min read


USFDA MD Guidance: Use of RWE to Support Regulatory Decision-Making forMedical Devices
Recently (14 December 2023) the U.S. Food and Drug Administration (FDA) Center for Biologics Evaluation and Research Center for Devices...

Sharan Murugan
Dec 19, 20232 min read


Philippines FDA: Guidance on Use of FDA eServices Portal for the Initial Application of CMDN
On 4 Decemberber 2023, the Philippines FDA released draft guidelines on the "Use of the Food and Drug Administration eServices Portal...

Sharan Murugan
Dec 6, 20232 min read


UK MHRA: Guidance on Register Medical Devices to Place on the Market
Last Friday on 1st December 2023 UK's Medicines and Healthcare Products Regulatory Agency (MHRA) released updated guidance on "Register...

Sharan Murugan
Dec 3, 20231 min read


Australia TGA: Clinical Evidence Guidelines
The Australian Regulatory Guidelines for Medical Devices (ARGMD) released updated guidance "Clinical Evidence" on 23 November 2023,...

Sharan Murugan
Nov 26, 20232 min read


Swiss Medic: Biosimilar Authorisation Guidance
Swissmedic, the Swiss Agency for Therapeutic Products, published an updated guidance on (26 November 2023) the "Guidance document...

Sharan Murugan
Nov 26, 20232 min read


South Africa's (SAPHRA): Questions And Answers Licensing Of Medical Device Establishments
Last Friday (17 November 2023) the South African Health Products Regulatory Authority (SAPHRA) released updated guidance on "Guideline On...

Sharan Murugan
Nov 19, 20231 min read


USFDA MD Guidance: Notifying FDA of a Permanent Discontinuance, 506J Guidance & Computational Model
USFDA's Center for Devices and Radiological Health and Center for Biologics Evaluation and Research released multiple guidances related...

Sharan Murugan
Nov 19, 20232 min read

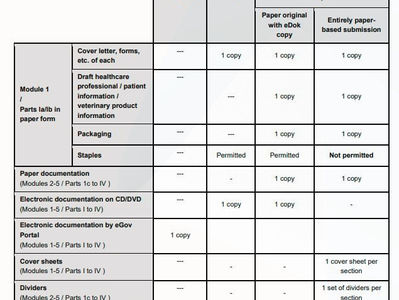
Switzerland's SwissMedic: Guidance on Formal Requirements
On 15 November 2023, Switzerland's Swissmedic released an updated guidance on "Formal Requirements" which serves as a resource for...

Sharan Murugan
Nov 15, 20232 min read


MHRA Guidance: Common Issues Identified during Clinical Trial Applications
Yesterday (06 November 2023) the UK's Medicines and Healthcare Products Regulatory Agency (MHRA) released updated guidance on "Common...

Sharan Murugan
Nov 7, 20231 min read


USFDA MD Guidance: Enforcement Policy for Clinical Electronic Thermometers
Last Friday (03 November 2023) the U.S. Food and Drug Administration (FDA) released two draft guidance "Enforcement Policy for Clinical...

Sharan Murugan
Nov 5, 20231 min read


USFDA Guidance: Supplements for Approved Premarket Approval or Humanitarian Device Exemption
Today (02 November, 2023) the U.S. Food and Drug Administration (FDA) Center for Biologics Evaluation and Research Center for Devices and...

Sharan Murugan
Nov 2, 20231 min read


TGA Guidance: General Dossier Requirements
Recently on 27th October, 2023 Australia's Therapeutic Goods Administration (TGA) released an updated guidance on the "General Dossier...

Sharan Murugan
Oct 31, 20231 min read


UK MHRA: Guidance on Software and Artificial Intelligence (AI) as a Medical Device
Earlier today (25 October 2023) UK's Medicines and Healthcare Products Regulatory Agency (MHRA) released updated Guidance on "Software...

Sharan Murugan
Oct 25, 20231 min read


USFDA MD Guidance: Enforcement Policy for Non-Invasive Remote Monitoring Devices
The U.S. Food and Drug Administration (FDA) Center for Devices and Radiological Health released a final guidance "Enforcement Policy for...

Sharan Murugan
Oct 23, 20231 min read


USFDA MD Guidance: Submission of Premarket Notifications for Magnetic Resonance Diagnostic Devices
Recently (10th October 2023) the U.S. Food and Drug Administration (FDA) Center for Devices and Radiological Health released multiple...

Sharan Murugan
Oct 14, 20231 min read


USFDA MD Guidance: AST System Devices, Technical Considerations & Electronic Submission Template
Recently (29th September 2023) the U.S. Food and Drug Administration (FDA) Center for Devices and Radiological Health released multiple...

Sharan Murugan
Oct 2, 20232 min read


USFDA MD Guidance: Cybersecurity-Quality System Considerations & Content of Premarket Submissions
The U.S. Food and Drug Administration (FDA) Center for Devices and Radiological Health released a final guidance "Cybersecurity in...

Sharan Murugan
Oct 2, 20231 min read


EC Q&A Guidance: Questions & Answers: Clinical Trials Regulation
Recently this Friday (29-Sepetember, 2023), the European Commission's Clinical Trials Coordination and Advisory Group released an updated...

Sharan Murugan
Oct 2, 20232 min read
