Search


EMA EMA 2025 AI Observatory Report: Artificial Intelligence in Medicines Regulation
Artificial Intelligence (AI) is rapidly becoming an integral part of pharmaceutical development and regulatory operations. From drug discovery and clinical trials to manufacturing and pharmacovigilance, AI is transforming how medicines are developed, assessed, and monitored. The European Medicines Agency (EMA) published its 2025 AI Observatory Report to provide an overview of AI-related activities across the European Medicines Regulatory Network (EMRN). The report highlights

Sharan Murugan
1 day ago3 min read


USFDA Press Release: AI Capabilities with Elsa 4.0 and New HALO Data Platform
Artificial intelligence (AI) is rapidly becoming an important part of healthcare, drug development, and regulatory operations. To modernize its internal systems and improve operational efficiency, the U.S. Food and Drug Administration (FDA) announced major upgrades to its AI infrastructure and data management capabilities. On May 6, 2026, the FDA announced the launch of Elsa 4.0, an upgraded version of its internal AI tool, along with the completion of a large-scale data plat

Sharan Murugan
May 103 min read


AI Announcement: Introducing GPT-Rosalind for Life Sciences Research
The global life sciences landscape is rapidly evolving with the integration of advanced artificial intelligence to accelerate drug discovery, genomics, and translational research . OpenAI has introduced a specialized model designed to address the complexity and scale of modern biomedical research workflows. The announcement Introducing GPT-Rosalind for life sciences research , published in April 2026 , presents a purpose-built AI model aimed at supporting scientific reasoning

Sharan Murugan
Apr 203 min read


EMA Guidance: Implementation of ICH E2D(R1) for Post-Approval Safety Reporting and the EU Network Data & AI Strategy
Regulatory authorities across the European Union continue to strengthen pharmacovigilance systems and data-driven regulatory decision-making. Two important EMA documents highlight these developments: the EU implementation strategy for the ICH E2D(R1) guideline on post-approval safety data and the Network Data Steering Group (NDSG) Workplan 2026–2028 on data and artificial intelligence in medicines regulation. Guidance: Network Data Steering Group Workplan 2026–2028: Data and

Sharan Murugan
Mar 154 min read


From Textbook Pages to an Interactive Periodic Table 📚⚡
I still remember sitting in the back of my chemistry class, flipping through massive textbooks just to compare atomic weights or understand a single element. A single question often meant turning dozens of pages. It was slow, and honestly, it sometimes took the excitement out of learning. Fast forward to today, and learning science has completely transformed. Instead of digging through hundreds of pages, we can now explore information instantly with interactive tools. That’s

Sharan Murugan
Mar 152 min read


USFDA Guidance: Use of Artificial Intelligence To Support Regulatory Decision & Guiding Principles of Good AI Practice in Drug Development
Artificial Intelligence (AI) is no longer a future concept in pharmaceutical research—it is already influencing how drugs and biological products are discovered, developed, manufactured, and monitored throughout their lifecycle. However, when AI outputs are used to support regulatory decisions , questions of trust, transparency, and accountability become critical. Recognising this, global regulators have begun defining expectations for the responsible use of AI in drug devel

Sharan Murugan
Jan 313 min read


SAHPRA Communication: Regulatory Requirements for AI/ML-Enabled Medical Devices in South Africa
Artificial Intelligence (AI) and Machine Learning (ML) are transforming healthcare globally, offering powerful tools for diagnosis,...

Sharan Murugan
Aug 23, 20252 min read


USFDA Guidance: Predetermined Change Control Plans (PCCPs) for AI/ML Medical Devices
Artificial Intelligence and Machine Learning (AI/ML) are increasingly used in medical devices—from diagnostic imaging software to digital...

Sharan Murugan
Aug 19, 20252 min read


European Commission Insights: Study on the Deployment of Artificial Intelligence in Healthcare – 2025
On 8 August 2025, the European Commission released an in-depth study on the " Deployment of artificial intelligence (AI) in healthcare...

Sharan Murugan
Aug 10, 20252 min read


European Commission: Guidelines on the Scope of the Obligations for General-Purpose AI Models established by AI Act
On 18 July 2025, the European Commission published detailed Guidelines on the scope of the obligations for providers of general-purpose...

Sharan Murugan
Jul 19, 20253 min read

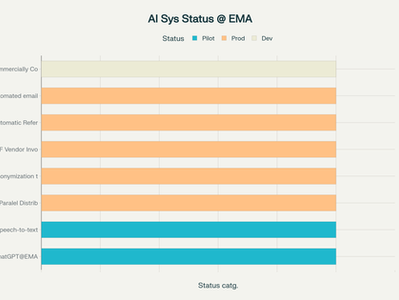
EMA’s AI Journey: The Rise of Artificial Intelligence in Medicines Regulation
In a world where artificial intelligence (AI) is moving rapidly from theory to practice, regulators are tasked with keeping pace—not only...

Sharan Murugan
Jul 19, 20253 min read


EMA NDSG Work Plan: Inside the Network Data Steering Group Workplan 2025–2028
In an era where data drives decisions and innovation defines progress, the European Medicines Agency (EMA) and Heads of Medicines...

Sharan Murugan
Jul 2, 20253 min read


UK MHRA News: "AI Airlock" Pioneering Safe Healthcare Innovation in the UK
The UK’s Medicines & Healthcare products Regulatory Agency (MHRA) has launched Phase 2 of its AI Airlock programme , supported by a...

Sharan Murugan
Jun 23, 20252 min read


Meet ELSA: USFDA Launches Agency-Wide AI Tool to Optimize Performance
What if the agonizing wait for drug approvals could be slashed from days to minutes? Imagine a future where breakthrough therapies reach...

Sharan Murugan
Jun 2, 20252 min read


EMA Network Data Steering Group workplan 2025-2028: Leveraging Data and AI for Enhanced Medicine Regulation
In the evolving landscape of medicines regulation, data and artificial intelligence (AI) have become pivotal tools to enhance public and...

Sharan Murugan
May 11, 20253 min read


Announcing Veeva AI: A Bold Leap Toward Intelligent Automation in Life Sciences
The world of pharma and life sciences is abuzz with a major announcement: Veeva Systems has just unveiled Veeva AI , a groundbreaking...

Sharan Murugan
Apr 29, 20253 min read


EMA's New AI Work Plan: Shaping the Future of Medicines Regulation
The European Medicines Agency (EMA) has unveiled a comprehensive Artificial Intelligence (AI) Work Plan to guide the integration of AI...

Sharan Murugan
Mar 31, 20252 min read


USFDA Guidance: Artificial Intelligence-Enabled Device Software Functions – Lifecycle Management and Marketing Submission Recommendations
The U.S. Food and Drug Administration (FDA) has issued a draft guidance document titled “ Artificial Intelligence-Enabled Device Software...

Sharan Murugan
Jan 7, 20252 min read


USFDA Draft Guidance: Considerations for the Use of Artificial Intelligence to Support Regulatory Decision
The U.S. Food and Drug Administration (FDA) released a draft guidance earlier today (07 January, 2025) outlining " Considerations for...

Sharan Murugan
Jan 7, 20252 min read


USFDA Final Guidance: Marketing Submission Recommendations for a Predetermined Change Control Plan for Artificial Intelligence-Enabled Device Software Functions
The US Food and Drug Administration (USFDA) has issued its final guidance on " Marketing Submission Recommendations for a Predetermined...

Sharan Murugan
Dec 9, 20242 min read
